DNA gives clues to clean and effective energy production
Researchers from several universities, including Uppsala University, have, by studying helical molecules in the body such as DNA, received clues as to how energy production in fuel cells can be made more efficient without admixture of carbon dioxide and heavy metals. The new research shows how the helical molecules, found naturally in all oxygen-dependent life, catalyses energy production in the cells.
Oxygen is important for a number of processes in the body, for example in cellular respiration, where glucose is transformed into water and carbon dioxide with the help of oxygen while simultaneously releasing energy: C6H12O6 + 6O2 –> 6H2O + 6CO2 + energi (ATP).
The challenge, however, is that glucose, water and carbon dioxide molecules assume what are known as ‘singlet’ states, while the oxygen assumes ‘triplet’ states, which means that the reaction cannot take place without adding anything to the chemical reaction. However, this can be done with the help of some form of catalyst.
Helical molecules may act as catalyst
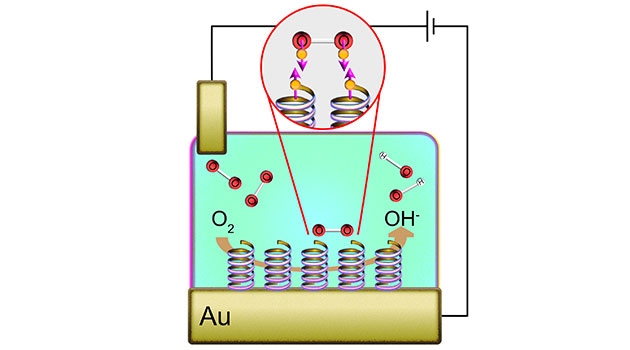
When it comes to other types of reactions, for example in fuel cells where hydrogen and oxygen are transformed into water to extract energy, heavy metals such as platinum may act as a catalyst. But so far it has not been known what the corresponding catalyst could be in the body, nor why this is done with such efficiency as it actually is.
Now, Jonas Fransson, Professor at the Department of Physics and Astronomy at Uppsala University has, together with chemists at the Weizmann Institute in Israel and the University of Pittsburg, USA, arrived at the conclusion that the unknown catalyst is likely to consist of helical molecules.
Cheap and effective fuel cells without heavy metals
The helical molecules are found in abundance in the body in the form of DNA and other polymers, and act catalytically for the type of reactions involving oxygen. Thanks to the helical molecules, the combined properties of the triplets and the singlets, such as energy and angular momentum, can be preserved during the chemical reactions where oxygen is broken down into oxygen atoms.
Physics and Astronomy, Uppsala University.
Photo: Mikael Wallerstedt
“For the reduction of oxygen to take place with great efficiency in the reaction, all biological helical molecules must be screwed in the same direction, which leads to an almost one hundred percent probability that the oxygen is taken care of in the body,” explains Jonas Fransson.
What makes the helical molecules efficient as catalysts is that they can split the oxygen into oxygen atoms which may react with glucose to produce energy, water and carbon dioxide in a completely different way than for example platinum. This means that it could be possible in the future to construct cheap and effective fuel cells without heavy metals.
Camilla Thulin
Publication
Yutao Sang, Francesco Tassinari, Kakali Santra, Wenyan Zhang, Claudio Fontanesi, Brian P. Bloom, David H. Waldeck, Jonas Fransson and Ron Naaman: Chirality enhances oxygen reduction, PNAS, DOI:10.1073/pnas.2202650119